 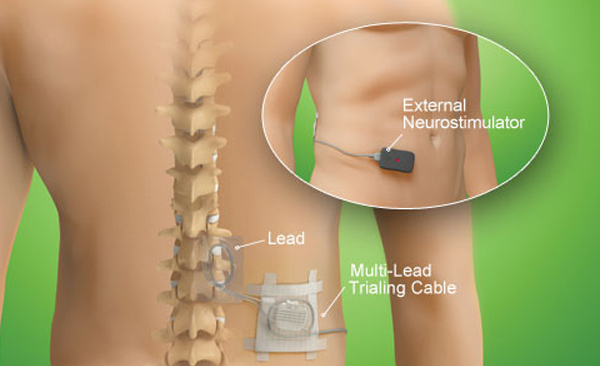
Its small size also allows for a smaller incision, which gives physicians increased flexibility in selecting the implant location. The thin profile and greater implant depth potentially makes the neurostimulator less noticeable and more comfortable for patients. Additionally, the device has the greatest recommended implant depth of any rechargeable SCS device. Slightly larger than the typical man’s watch, the Eon Mini neurostimulator has a thin 10 mm profile and weighs 29 grams (approximately 1 oz). The neurostimulator, which is both FDA cleared and CE marked, has the best battery life and lowest physical profile among rechargeable SCS devices. Jude Medical has announced Japanese approval of its Eon Mini implantable spinal cord stimulation (SCS) device for treatment of chronic pain in the lower back or legs. Similar to FBSS, post-surgical pain is chronic pain that occurs after surgery. These include the following: Post-surgical pain. Apart from FBSS, there are other medical conditions that can benefit from alternative pain management treatments. The Eon Mini is the smallest, rechargeable implantable pulse generator on the market. It uses microelectronic circuitry to generate constant current electrical stimulation. Implantable neurostimulator / medullary Eon Mini St. It is powered by a hermetically sealed battery within a titanium case. Vederman, Aaron Viswanathan, Ashwin Watson, C. A dorsal column stimulator can provide pain relief for more than one condition. The Eon Mini Implantable Pulse Generator (IPG) is a 16-channel, rechargeable, multi-program system that allows the connection of one or two leads with four, eight, and 16 electrode leads. Jude Medical 8 17.3 Pc 8 NA Medtronic 8 < 33b Pc 9 Tripole St. Tavanaiepour, Daryoush Vasudevan, Sridhar V. Jude Medical 4 10 Pc 6 NA Boston Scientific (Advanced Bionics) 8 16.7 L 7 3186 St. Penhollow, Tammy Pilitsis, Julie Portenoy, Russell K. Dade Luther, Neal Maarrawi, Joseph Marcus, Norman McCartney, Shirley Mertens, Patrick Meyerson, Björn A. 
Linderoth, Bengt Liu, Chang-Chia Loeser, John D. Lang, Shih-Shan Lee, Jung-Il Lee, John Y. Although more than 50 pain relief could be achieved with initial SCS using a Tripole 8 3-column paddle lead (St. Hussain, Arif Ingram, Susan Kalia, Suneil K. A Dorsal Column Stimulator (DCS) is a medical device used to manage chronic pain by delivering electrical stimulation to the dorsal column of the spinal cord. Griffith, Adam Hamani, Clement Harsh, Viraat Heinricher, Mary M. Göçmen, Selçuk Gonzalez-Sotomayor, Julio A. Chen, Grace Cheyuo, Cletus Chou, Roger Cleary, Daniel R. Barbaro, Nicholas Barolat, Giancarlo Bascom, Paul Belzberg, Allan J. at ¶ 15) On March 4, 2005, the FDA issued an approval for the commercial distribution of the Eon Neurostimulation System, Model 3716. Since the problem surfaced in 2011, over 300 heat-related complaints have been filed on behalf of carriers of defective units and several lawsuits have been filed. Jude’s Genesis and Eon family of Neurostimulation (IPG) Systems. Physicians and Patients who may have been implanted with defective stimulators are being notified individually by St. St Jude Medical is implementing a design change to the charging systems to prevent excessive heating while charging. November 21, 2001, the FDA issued an approval for the commercial distribution of St. Abbott, Elizabeth Emily Acar, Feridun Ahmed, Ebtesam Ajuwon, Abideen A. As of 30 June 2012, the worldwide rates of reporting of excessive heating while charging for each of the models remain relatively low (Eon 0.44, Eon Mini 0.47 and Brio 0.21 of devices implanted). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
